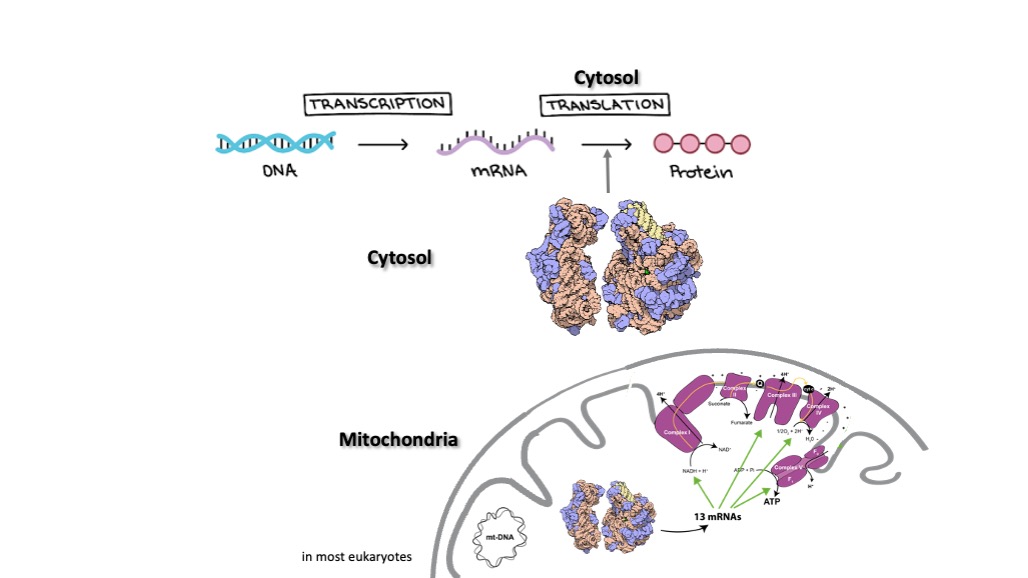
Mitochondria usually possess their own genome and translation apparatus. Kinetoplastids for instance, such as trypanosomes, count a single mitochondrion where the mitochondrial DNA is called the kinetoplast (k-DNA) and consists of concatenated circular DNA molecules, made of maxicircles and minicircles. The maxicircle DNA codes for the two rRNAs of relatively short sizes (9S and 12S) and 18 mRNAs. 12 mRNAs out of these 18 undergo unique and extensive editing thanks to the minicircles-encoded guide RNAs by a macromolecular complex called the editosome. Once the mitochondrial mRNAs (mt-mRNAs) are edited and matured, they are translated by the mitoribosome. The mitoribosome is a hybrid molecule, made of mitochondrion-encoded rRNAs and mostly nuclear-encoded r-proteins. Most of mt-mRNAs code for components of the respiratory chain complexes (including the ATP synthase).
Kinetoplastids are mainly heteroxenous, with different morphological forms depending on the life cycle stage. The global life-cycle is similar amongst kinetoplastids, which spend most of their life in the guts of an insect vector (e.g. procyclic form for T. brucei) before transmission to the host often upon biting, where they differentiate to a replicative bloodstream form. Consistently, large-scale morphological differences in the single mitochondrion are reported between the insect-host and the vertebrate-host forms of the parasites. In T. brucei for instance, the procyclic form has a respiring mitochondrion that produces ATP via the ATP synthase while the bloodstream form is characterized by a single long tubular mitochondrion with a down-regulated respiratory chain that hydrolyses ATP via the same ATP synthase molecule; i.e., the ATP synthase works reversely between these two forms of the parasites. The ATP synthase is located in the inner mitochondrial membrane and its oligomerization additionally controls the cristae formation and the shape of the mitochondrion. Thus, the ATP synthase from is essential for the parasite’s survival.